|
3/2/2023 0 Comments Atomic orbitalsFor more rigorous and precise analysis, the numerical approximations must be used.Ī given (hydrogen-like) atomic orbital is identified by unique values of three quantum numbers: 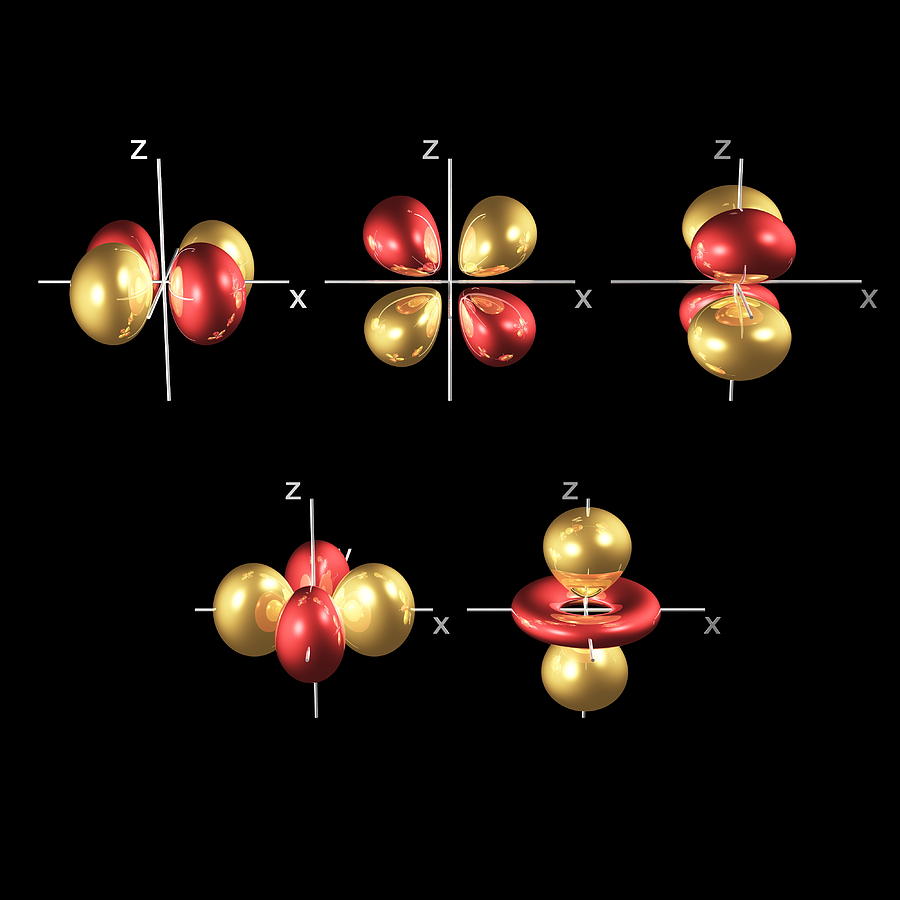
Orbitals of multi-electron atoms are qualitatively similar to those of hydrogen, and in the simplest models, they are taken to have the same form. An atom of any other element ionized down to a single electron is very similar to hydrogen, and the orbitals take similar forms.įor atoms with two or more electrons, the governing equations can be solved only by using the methods of iterative approximation. The simplest atomic orbitals are those that occur in an atom with a single electron, such as the hydrogen atom. An orbital can be described as a "wave function" of an electron in an atom, and the shape of an orbital indicates the probability of locating the electron within a particular region of the atom. Each atomic orbital has a characteristic energy level and a particular distribution of electron density. In quantum mechanics, atomic orbitals are the quantum states (or discrete energy states) that electrons surrounding an atomic nucleus may exist in. 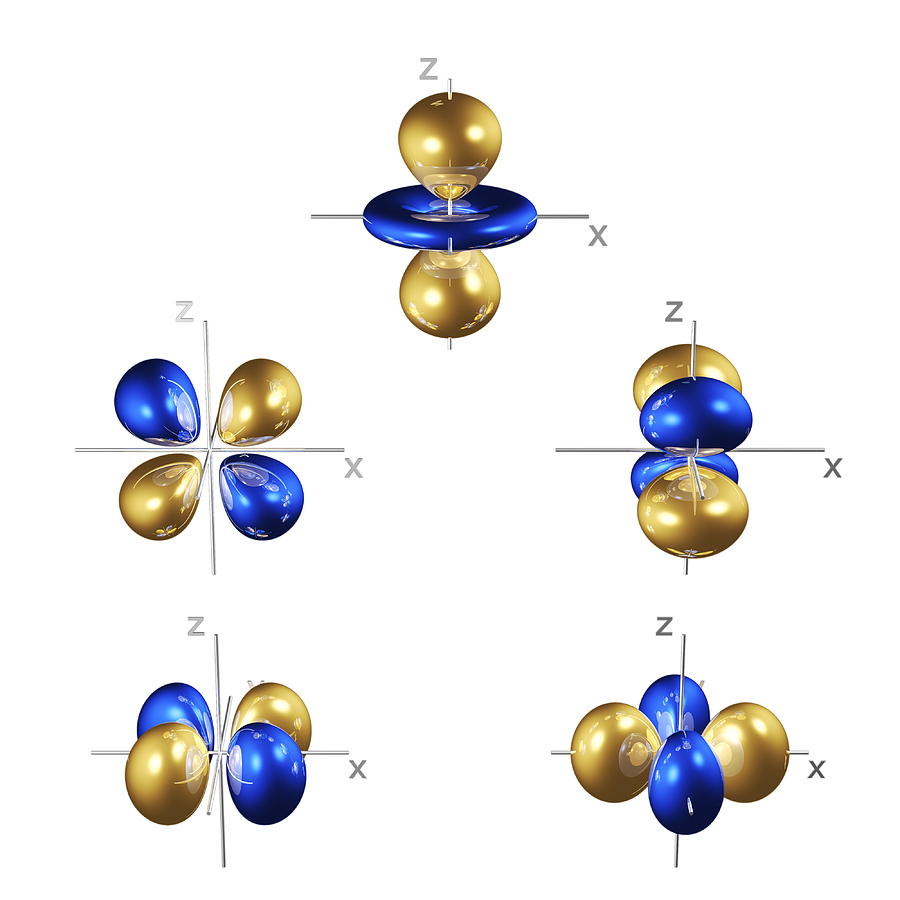
Atomic orbitalsĮxplaining the distribution and behavior of electrons within an atom was one of the driving forces behind the development of quantum mechanics. For this reason, the term orbit was replaced by the term orbital. Consequently, the electron was thought of as a "cloud" distributed around a nucleus, like a large atmosphere around a tiny planet. As the concept of an electron shifted from a solid particle to an entity with both wave-like and particle-like properties, it became clear that the electron does not have a well-defined position or orbit within the atom. In the classical model, electrons were thought to orbit the atomic nucleus much like planets orbiting the Sun (or moths orbiting speedily around a lamp).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
